A simple way to determine strength is to add the acid or base to water—high reactivity means a stronger acid or base This reactivity is measured with a value called the \(K_a\) or \(K_b\) value It tells us how reactive an acid or a base is based on how many hydronium or hydroxide ions are formed during the reactionDefine a strong and a weak acid and base Recognize an acid or a base as strong or weak Determine if a salt produces an acidic or a basic solution Except for their names and formulas, so far we have treated all acids as equals, especially in a chemical reaction However, acids can be very different in a very important way Consider HCl(aq) When HCl is dissolved in H 2 O, itExperts are tested by Chegg as specialists in their subject area We review their content and use your feedback to keep the quality high

What Are Strong Acids Or Bases Are That Ionize Or Dissociate In Aqueous Solution Socratic
Is hi a strong or weak acid
Is hi a strong or weak acid- Acid dissociation constant gives an indication of the ability to lose a proton of a weak acid To check whether an acid is strong or weak we can identify with the help of various indicators like litmus paper or pH paper In the pH scale, from 16 acids are represented An acid with pH 1 is said to be very strong, and as the pH value increases, acidity is decreasedWeak and strong should not be mistaken for dilute and concentrated A dilute acid has the acid molecules mixed with a large amount of water, so that there is only a low concentration of H ions




Classify Each Substance As A Strong Acid Clutch Prep
Strong AcidWeak Base and Weak Acid Strong Base CH 3 COOH(aq) OH(aq) > CH 3 COO(aq) H 2 O(l) Slow change in p H before equivalence point;It is perfectly possible to have a concentrated solution of a weak acid, or a dilute solution of a strong acid Read on Strong acids Explaining the term "strong acid" We are going to use the BronstedLowry definition of an acid Note If you don't know what the BronstedLowry theory of acids is, you should read about theories of acids and bases on another page in this section YouStrong base weak acid Example 7 Write the balanced chemical equation for the dissociation of Ca(OH) 2 and indicate whether it proceeds 100% to products or not Solution This is an ionic compound of Ca 2 ions and OH ions When an ionic compound dissolves, it separates into its constituent ions Ca(OH) 2 > Ca 2 (aq) 2OH (aq) Because Ca(OH) 2 is listed in Table122, "Strong
HOW CAN U TELL IF HNO3 KNO3 IS A BUFFER SOLUTION A buffer solution must contain a weak acid and its conjugate base OR a weak base and its conjugate acid HNO3 is a strong base and KNO3 is the salt of a strong base (KOH) and a chemistry Strong base is dissolved in 665 mL of 0400 M weak acid (Ka = 369 × 105) to make a buffer with a pH of Is HI a strong or weak base?See the answer See the answer See the answer done loading Show transcribed image text Expert Answer Who are the experts?
Strong or Weak Acid or Base study guide by BRoemer21 includes 24 questions covering vocabulary, terms and more Quizlet flashcards, activities andAnswer and Explanation 1 Hydroiodic acid, HI H I , is one of seven strong acids, which means that it completely ionizes in water to produce 1 mole of hydronium ions for every mole of acid dissolved To check If Nitric acid(HNO 3) a strong or weak, first we have to take a clear understanding of the differences between strong and weak acids A strong acid is generally a compound that dissociates completely or is 100% ionized in a solution to yield H ions which means no undissociated acid remains in the solution, all moles of acid completely break off and release




Why Hydrogen Fluoride Is A Weaker Acid Than Hydrochloric Acid Find Acidic Order Of Hf Hcl Hbr Hi Youtube




Packet 10 Acids Bases And Salts Reference Tables K L M J T Ppt Download
Identify HClO as a weak acid,strong acid,weak base,strong base both weak acid and weak base or neither acid or base There are six common stron acids HNO3,HClO4,H2SO4,HCL,HBr,HI and strong base are all the group Ist hydroxides and the Three of Strength of Conjugate Acids and Bases Chemistry Tutorial Strong Acid – Weak Conjugate Base Pair A strong BrønstedLowry acid There are two ways to determine whether HI is a strong or weak acid The first it to memorize the seven common strong acids For general chemistry courses t Solution A is a strong acid, solution B is a weak acid, solution C is a strong alkali asked in Class X Science by muskan15 Expert ( 379k points) acids bases and salts




Identify Salts As Neutral Acidic Or Basic Video Khan Academy
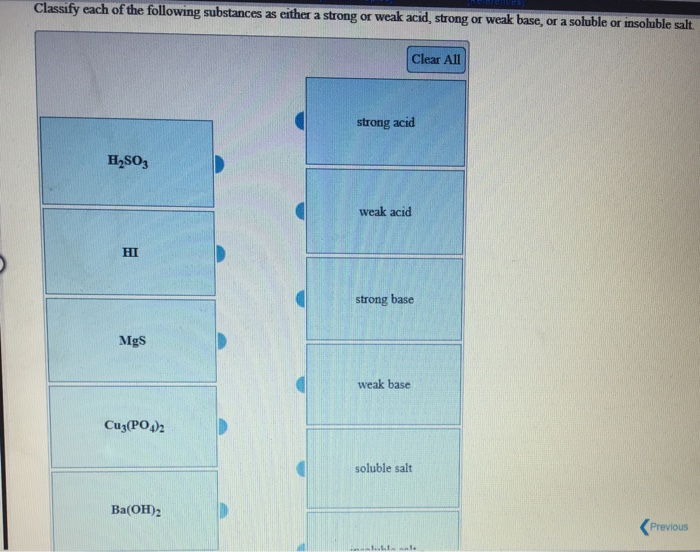



Solved Classify Each Of The Following Substances As Either A Chegg Com
HI KOH HNO 3 Mg(OH) 2 H 2 SO 4 Ca(OH) 2 HClO 4 By analogy, a strong base A base that is 100% ionized in aqueous solution is a compound that is essentially 100% ionized in aqueous solution As with acids, there are only a few strong bases, which are also listed in Table 102 "Strong Acids and Bases (All in Aqueous Solution)" If an acid is not listed in Table 102 "Strong Classify each substance as a strong acid, strong base, weak acid,or weak baseStrong Acid Question Classify each substance as a strong acid, strong base, weak acid,or weak baseStrong Acid This problem has been solved! So how do we know if an acid or base is strong or weak?
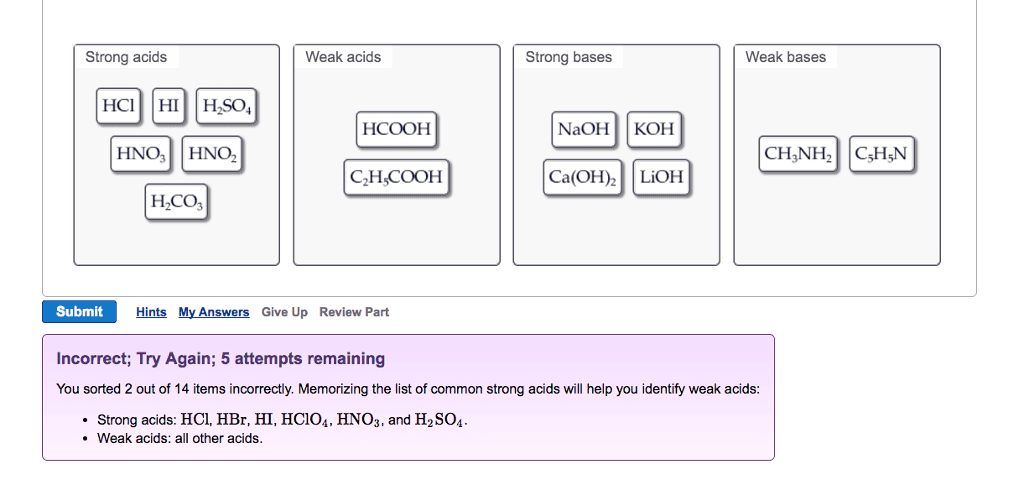



Oneclass Strong Acids Weak Acids Strong Bases Weak Bases Naoh Koh Hcooh Hno3 Hno2 Ch3nh2 C5h5n C2h C




I Need Help With Chemistry Homework For Acids And Bases Clutch Prep
Strong and Weak Acids and Bases 1 The following common acids are strong HCl, HBr, HI, HNO 3, HClO 4, H 2 SO 4 The following are some less common acids that are also strong HClO 3, HBrO 3, HIO 3, H 2 SeO 4 Assume all other acids are weak unless told otherwise Some weak acids HF, HNO 2, HClO 2, H 2 SO 3 = SO 2 H 2 O, HC 2 H 3 O 2 = HOAc 2 All ionic hydroxides are strong basesAnswer (1 of 7) This answer is only for Bronsted acids (proton donors), as I think that is what the OP is looking for Some answers mention the pH Others mention that the concentration dependence of the scale makes it an impractical choice 10^{7}M H_2SO_4 is less acidicSolution is a buffer CH 3 COOH(aq)/CH 3 COO(aq) At halfway point HA = A p H = p Ka At equivalence, p H determined by CH 3 COO(aq)




Acid Base Diagram Quizlet




Chapter 16 Acid Base Equilibria Ppt Download
Wiki User ∙ Add an answer Want this question answered?Where HA is a protonated acid, H is the free acidic proton, and A – is the conjugate base Strong acids yield weak conjugate bases For sulfuric acid, which is diprotic, the "strong acid" designation refers only to the dissociation of the first proton latex\text{H}_2\text{SO}_4 (\text{aq}) \rightarrow \text{H}^ (\text{aq}) \text{HSO}_4 ^ (\text{aq})/latex More precisely, theIf ANY acid has a high concentration then this will consequently increase the hydrogen ion concentration making the acid 'stronger' The 2 State whether a given acid or base is strong or weak Students should consider hydrochloric acid, nitric acid and sulfuric acid as examples of strong acids and carboxylic acids and carbonic acid (aqueous carbon dioxide) as weak



Acids




Acid Base Reactions Definitions Strong Acids Hcl Hbr Hi Hno 3 Hclo 4 H 2 So 4 Acid A Species That Supplies H Ions To Water Strong Acid Ppt Download
Mg(OH) 2 C 5 H 5 N;Acid strength is the tendency of an acid, symbolised by the chemical formula, to dissociate into a proton, , and an anion, The dissociation of a strong acid in solution is effectively complete, except in its most concentrated solutions Examples of strong acids are hydrochloric acid (), perchloric acid (), nitric acid and sulfuric acid () A weak acid is only partially dissociated, with Equilibrium constants of strong bases are high while those of weak bases are low, on the equilibrium scale, as the acid gets weak the value shifts to its left Strong bases are good electrolytes while weak bases are not Conclusion The tendency to dissociate ions defines the strength of a base Each strong base has a conjugate weak acid and a weak base has its conjugate strong acid Strong




Chapter 2 Acids Bases 2 Arrhenius Acids And Bases Bronstead Lowry Acids And Bases Acids And Bases Acid Base Systems Ppt Download




Is Hi An Acid Or Base Strong Or Weak Hydroiodic Acid
Be notified when an answer is posted 📣 Request Answer Add your answer Earn Is Baking Soda a Strong or Weak Base?In chemistry, neutralization or neutralisation (see spelling differences), is a chemical reaction in which an acid and a base react quantitatively with each other LIST ACID NH4ClO4 NH4Cl HBrO (WEAK) H2PO4H3PO3 (WEAK) HNO3 (STRONG) HCl (STRONG) H2S (WEAK) H2SO4 (STRONG) H3PO4 (WEAK) H2CO3 (WEAK) HBr (STRONG HI (STRONG) HClO4 (STRONG) HClO3
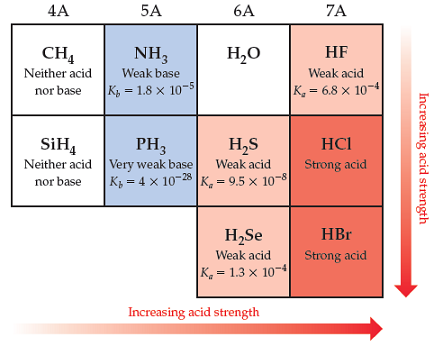



Solved Part A Are The Acid Properties Of Hi What You Chegg Com




Acid Base Properties Of Salts Video Khan Academy
There is no sharp boundary between weak and strong acid Wikipedia defines strong acids as acids which ionize completely in aqueous solution So, an acid, say $\ce{HA}$ , is said to be strong if one mole of this acid dissociates in aqueous solution toBaking Soda is a weak acid Baking soda is made from sodium bicarbonate, a chemical compound with the formula NaHCO3 Sodium bicarbonate (baking soda) is probably the most familiar alkali used in cooking It is actually a salt of a strong base and a weak acid so that it acts as a weak base Classify HI as a strong or weak acid or base Weak base strong acid strong base weak acid This problem has been solved!




Ppt Strong Acids Vs Weak Acids Powerpoint Presentation Free Download Id



3
See the answer See the answer See the answer done loading Classify each substance as a strong acid, strong base, weak acid,or weak base Strong Acid HBr HNO 3 HIAcids and bases come in pairs called conjugate acid/conjugate base When an acid loses a proton it forms its conjugate base When a base accepts a proton it forms its conjugate acid When strong acids are dissolved in water, they dissociate completely Examples include HCl, HBr, HI, HNO3, H2SO4, HClO4 When weak acids are dissolved in waterSolution Because HCl is listed in Table 121, it is a strong acid Because Mg(OH) 2 is listed in Table 121, it is a strong base The nitrogen in C 5 H 5 N would act as a proton acceptor and therefore can be considered a base, but because it does not contain an OH compound, it cannot be considered a strong base
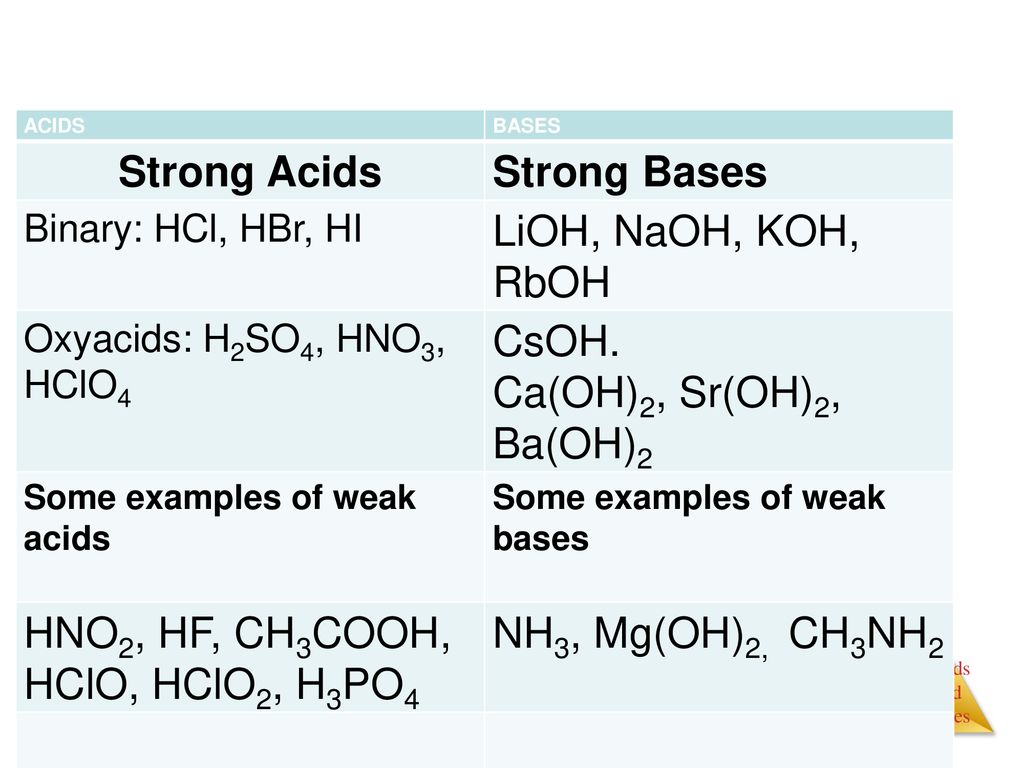



Chapter 16 Acids And Bases Ppt Download




Worksheet 22 A Salts Shown Below Is A Chart Of Acid Their Conjugate
Identify each acid or base as strong or weak HCl; Main Differences Between Strong Acid and Weak Acid Strong acids react faster whereas weak acids take time to react with any base Strong acids are good conductors of electricity on contrary weak acids are not too good as conductors of electricity Strong acid passes electricity faster whereas weak acids are slow conductorsLearn the surprising difference between strong and weak acids and bases Learn a new word that might relate to your latest




Mcat General Chemistry Acids And Bases Flashcards Quizlet



Solved So I M Pretty Sure I Have Hi Hcn Ba Oh 2 And Nh3 In The Right Places But I M Not Too Sure About H2so4 Csoh And H2so3 Can Someone Pleas Course Hero
Strong Acids and Weak Acids Classification Scheme based on pKa The dissociation of an acid HA is determined by its acidity constant K a (1) HA = H A with K a = H A / HA Strong acids dissociate completely in water, while weak acids do not dissociate completely A classification based on acidity constants or pK a values seemsWeak acids only partially dissociate, therefore the acid and its conjugate base will both be present at equilibrium The list of strong acids are as follows (commit these to memory) HCl hydrochloric acid, HNO3 nitric acid, HI hydroiodic acid, HBr hydrobromic acid, HClO4 perchloric acid, H2SO4 sulfuric acid All other acids are weak acids Weak acid and strong base For this case, the reaction is the following (with $\ce{AH}$ as the weak acid) $$\ce{AH(aq) OH(aq) > A(aq) H2O(l)}$$ The reaction enthalpy for this reaction depends on the nature of the weak acid Specifically, the answer depends on the difference in the enthalpy of formation of $\ce{A(aq)}$ vs $\ce{AH(aq)}$ Conceptually, it takes
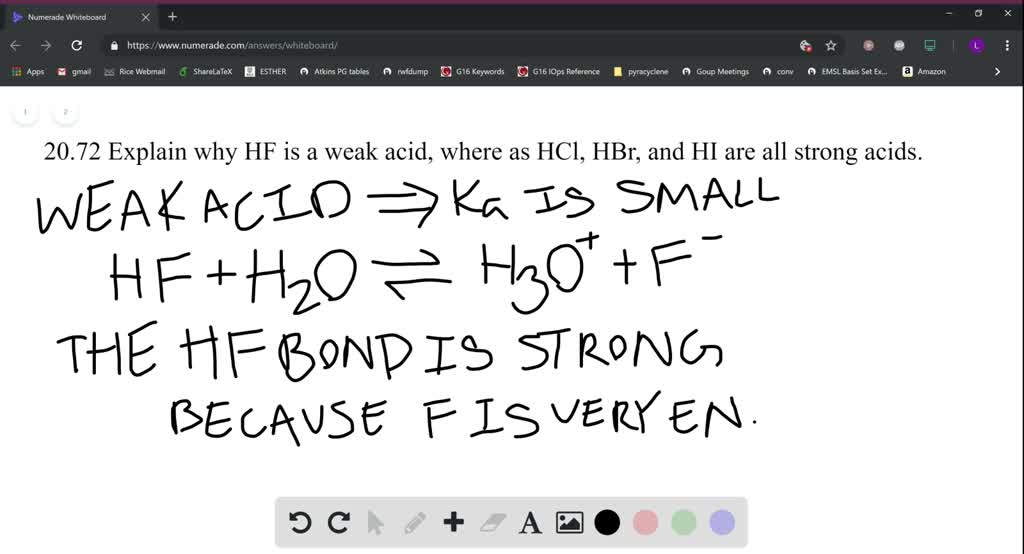



Solved Explain In Detail Why Hbr Is A Much Stronger Acid Than Hf To Do So Draw Acid Base Equilibria Reactions For Both Compounds And Compare The Relative Stabilities Of Their Conjugate Bases



1
What makes an acid strong or weak? No The strong acids are HCl, HBr, HI, HNO3, HClO3, H2SO4, and HClO4 Every other acid is a weak acidStrong acids have Ka>1, bases have Kb>1, Weak acids and bases have conjugates that are also weak acids or bases STUDY PLAY HCl Strong acid HBr Strong acid Hi Strong acid HF Weak acid HClO4 Strong acid H2SO4 Strong acid HNO3 Strong acid NaOH Strong base this includes all group one metals bound to hydroxide
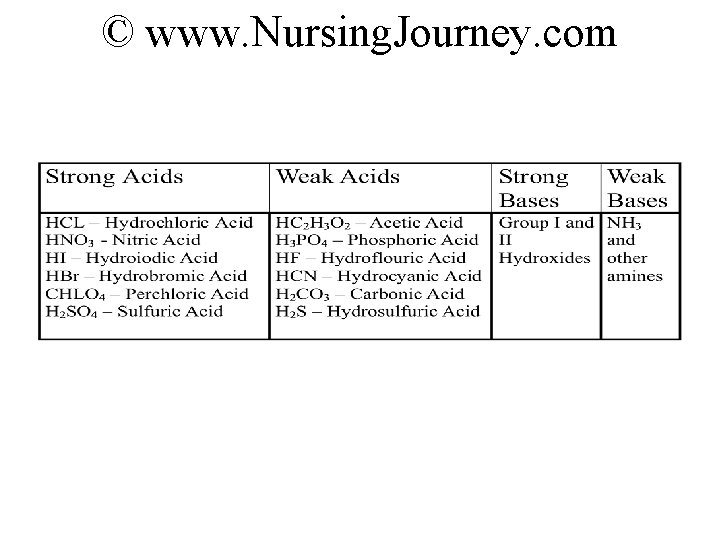



Www Nursing Journey Com Strong Or Weak Acids




Aleks Understanding The Difference Between Strong And Weak Acids Youtube
Answer (1 of 4) HBr is a strong acid Its pKa value is 9 and therefore it almost completely ionises to give a proton in aqueous form This is due to a weak covalent bond between H and Br atoms in the molecule, giving a strong ionic characteristic to the bonding Moveover, HI is even stronger t A weak acid is an acid that partially dissociates into its ions in an aqueous solution or water The conjugate base of a weak acid is a weak base, while the conjugate acid of a weak base is a weak acid At the same concentration, weak acids have a higher pH value than strong acids




Chemistry Guide The Quick Guide To Acids And Bases By Forum E Forum Education Guidebooks Medium



3




Strong Acids And Bases Cheat Sheet Study Guide Mcat And Organic Chemistry Study Guides Tutoring




Strong Acids And Bases Cheat Sheet Study Guide Mcat And Organic Chemistry Study Guides Tutoring




Topic 4 Acid Base Equilibria List Three Strong Acids And Three
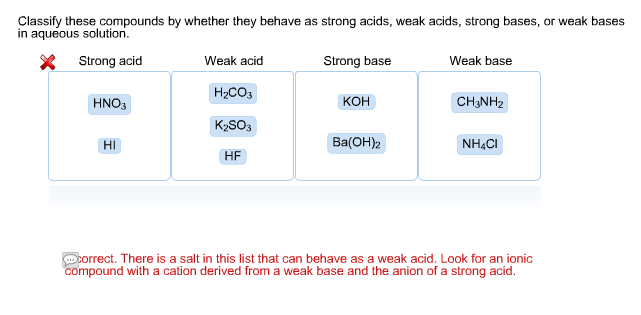



Solved Classify These Compounds By Whether They Behave As Chegg Com
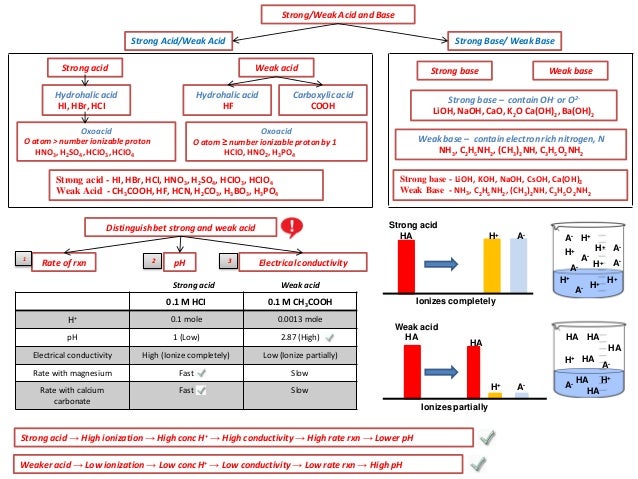



Ib Chemistry On Acid Base Buffers




Strong And Weak Acids And Bases




Lecture 10 The Strong Acid Strong Base Equilibrium Calculation
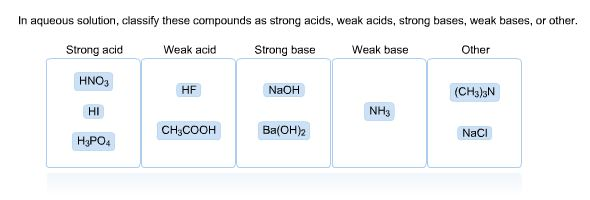



Solved In Aqueous Solution Classify These Compounds As Chegg Com
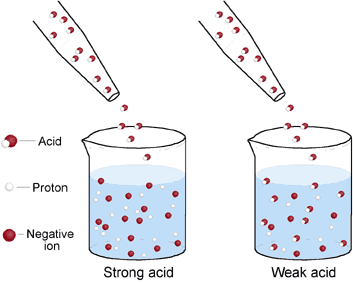



Acid Base




Solved Hcl Hbr And Hi Are Strong Acids However Hf Is A Chegg Com




Is Hi Hydroiodic Acid A Strong Or Weak Acid Youtube




List Of Strong Weak Acids Bases Chemistry Basics Chemistry Organic Chemistry




What Are Strong Acids Or Bases Are That Ionize Or Dissociate In Aqueous Solution Socratic
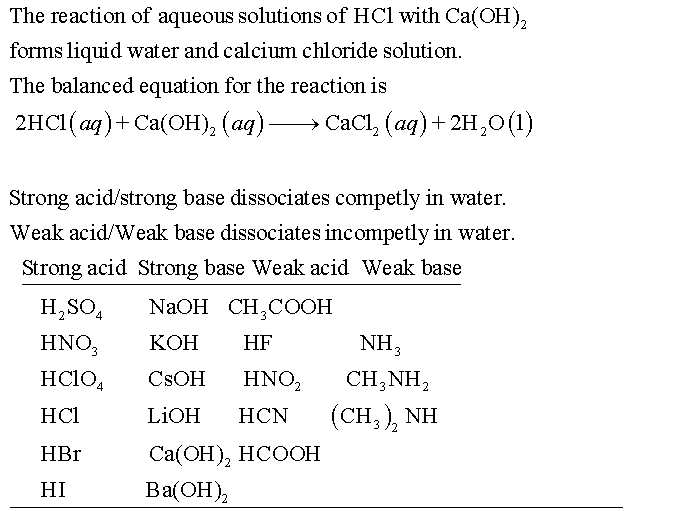



Classify Each Substance As A Strong Acid Strong Base Weak Acid Or Weak Base Home Work Help Learn Cbse Forum




Acids And Bases Chapters 14 15 Common Acids
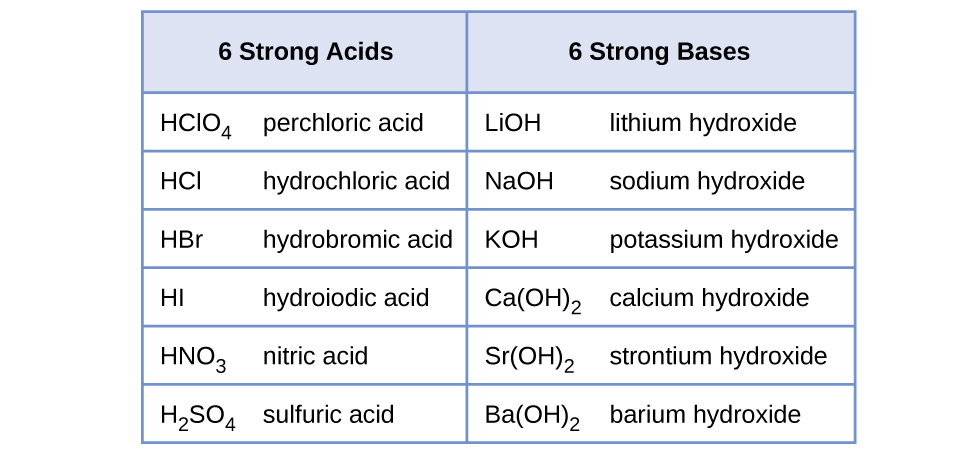



14 3 Relative Strengths Of Acids And Bases Chemistry




Mcat Mnemonics Strong Acids Prospectivedoctor




Strong Acids And Bases Overview Video Chemistry Ck 12 Foundation




Classify Each Substance As A Strong Acid Clutch Prep
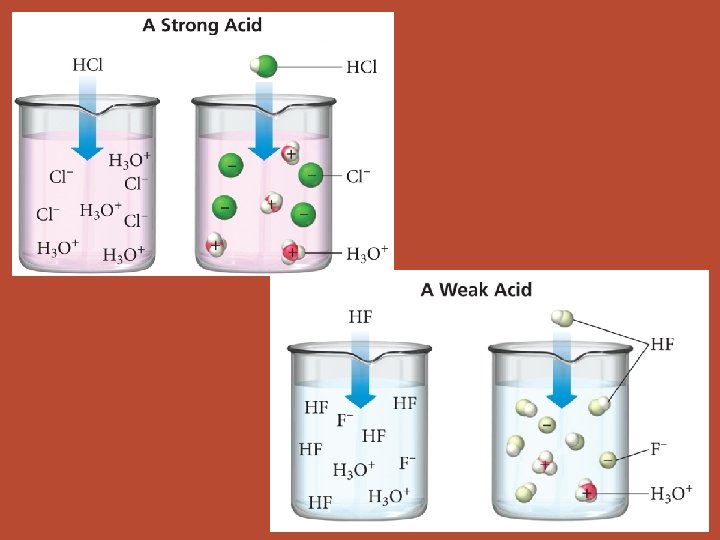



Acids And Bases Three Definitions Arrhenius Acid Produces




Strong And Weak Acids And Bases Numerade




Hno 3 Hcl Hbr Hi H 2 So 4 And Hclo 4 Are The Strong Acids Strong And Weak Acids Bases The Strength Of An Acid Or Base Is Determined By The Amount Ppt Download




Strong Acids And Bases Mcat Chemistry Cheat Sheet Study Guide Studypk Chemistry Lessons Chemistry Education Chemistry Classroom




Acids And Bases I Introduction




Chapter 2 Acids Bases Ppt Download




Strong And Weak Acids Bases Ppt Download




Strong Acids Vs Strong Bases Flashcards Quizlet



1



2




Strong And Weak Acids Bases Ppt Download




Is Hi An Acid Or Base Strong Or Weak Hydroiodic Acid




Strong Acid Solutions Video Khan Academy




Is Hi Hydroiodic Acid A Strong Or Weak Acid Youtube



What Are Strong And Weak Acids Example




Is Hbr A Strong Or Weak Acid Youtube
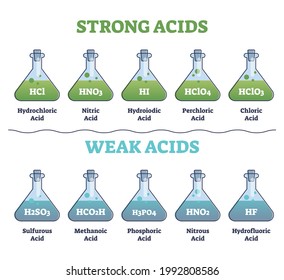



Strong Acid Images Stock Photos Vectors Shutterstock




Chem 2 Acid Base Equilibria Iv Calculating The Ph Of Strong Acids
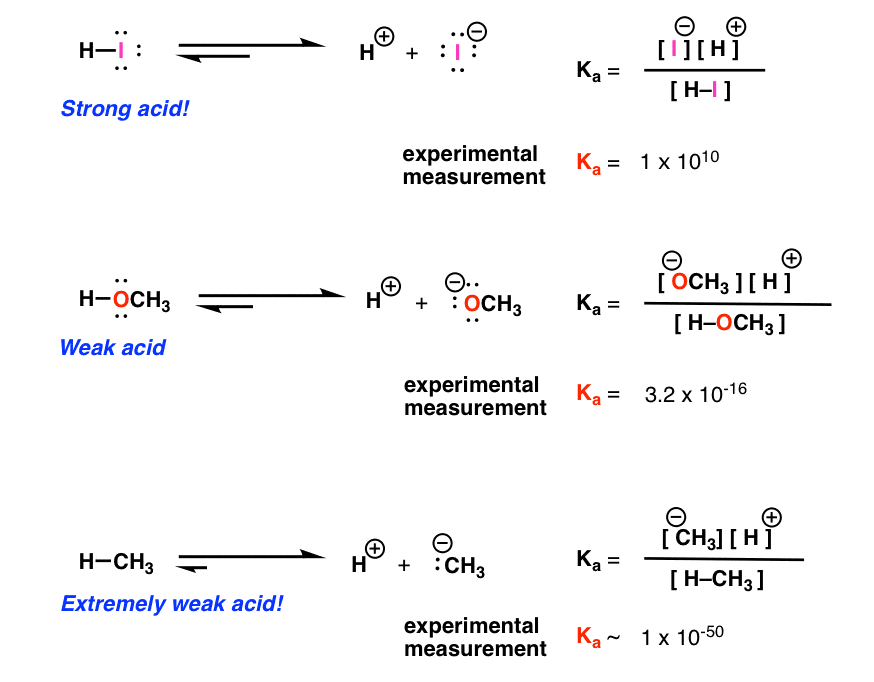



Acid Base Reactions Introducing Ka And Pka Master Organic Chemistry



Which Is The Strong And Weak Acid In These Hf Hcl Hbr And Hi Quora
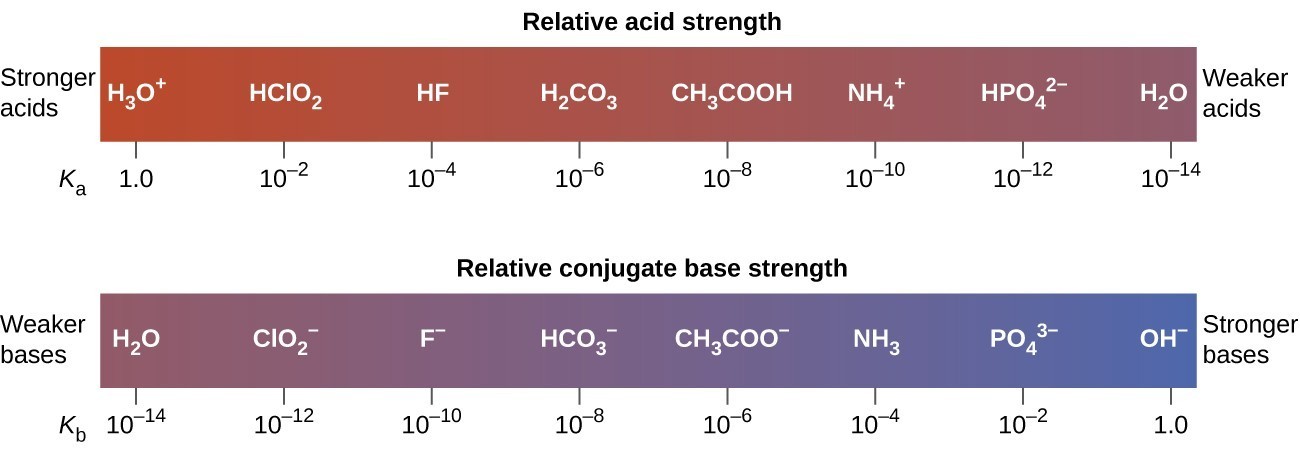



Relative Strengths Of Acids And Bases Chemistry For Majors




36 Which One Of The Following Is Not A Strong Acid Or A Weak Electrolyte A Hno Hci Hi Hf E Hcio Answer 37 The Is The Conjugate Species That Remains After




Is Hi An Acid Or Base Strong Or Weak Hydroiodic Acid




For The Following Compound Predict Whether The Solution Is Clutch Prep




Chapter 10 Acids Bases Characteristics Of Acids Sour
/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)



List Of Common Strong And Weak Acids
/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)



List Of Common Strong And Weak Acids



2



What Are Some Examples Of Strong And Weak Acids And Bases Quora
:max_bytes(150000):strip_icc()/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)



List Of Common Strong And Weak Acids



Difference Between Strong And Weak Acid Difference Between




Chemistry The Central Science Chapter 4 Section 3




Titration Of A Strong Acid With A Strong Base Chemistry Libretexts




Acids Bases




Solved Classify The Compounds As A Strong Acid Weak Acid Chegg Com




2 1 16 Strong And Weak Acids Pearson Schools




Super Trick To Learn Example Of Strong Acid Strong Base Weak Acid Weak Base Type Of Salt Ionic Youtube




Strong Weak Acids Bases Mr Carson S Science Page
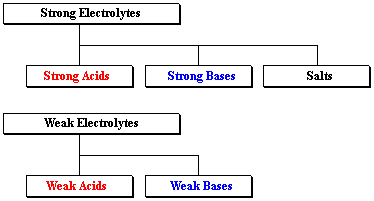



Classifying Electrolytes




Classify The Compounds As A Strong Acid Weak Acid Strong Base Or Weak Base Strong Acid Homeworklib



Solved Acidic And Basic Solutions Have Distinct Physical And Chemical Properties For Chemistry Courses Especially Those With A Laboratory Compone Course Hero




Classify Each Of The Following Substances As Either A Strong Clutch Prep




Get Answer Classify These Compounds As Strong Acids Or Weak Acids Strong Transtutors




8 3 Strong And Weak Acids And Bases
/Lithium-hydroxide-xtal-3D-SF-5897471f5f9b5874ee0edf7a.png)



Determining The Strength Of Acids And Bases
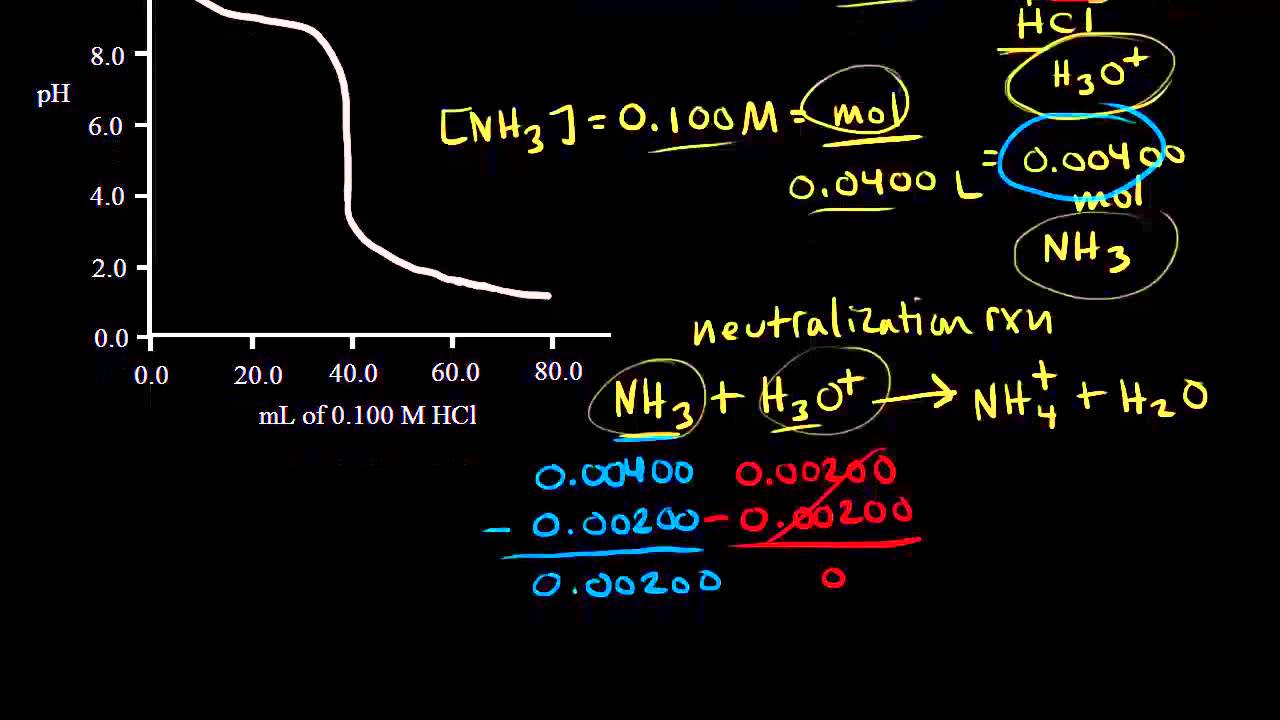



Titration Of A Weak Base With A Strong Acid Video Khan Academy




Strength Of Acids Boundless Chemistry




Is Hi A Strong Acid Techiescientist




Is Hi An Acid Or Base Strong Or Weak Hydroiodic Acid
/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)



List Of Common Strong And Weak Acids
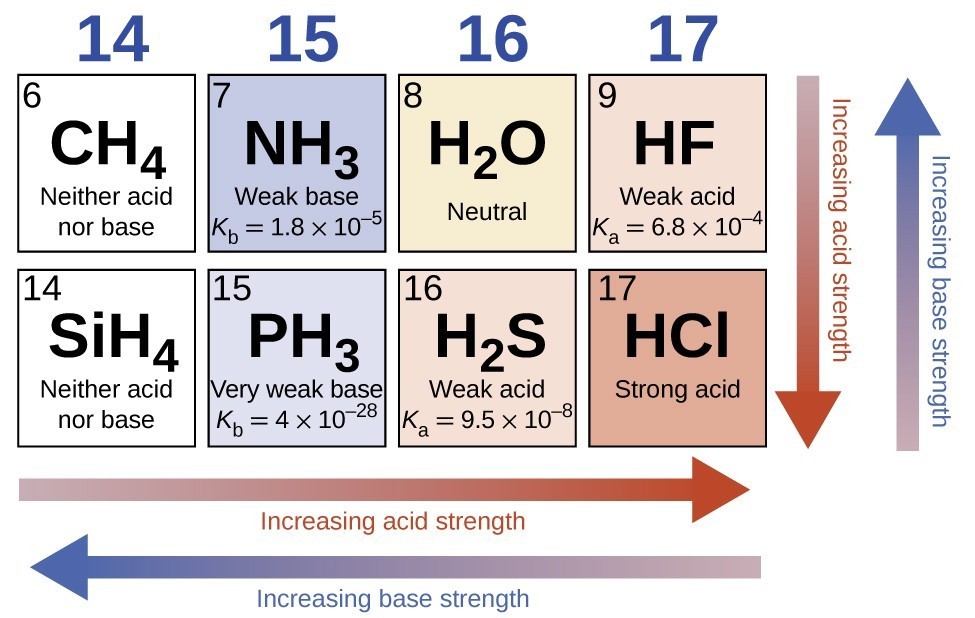



14 3 Percent Ionization And Relative Strengths Of Acids And Bases Chemistry
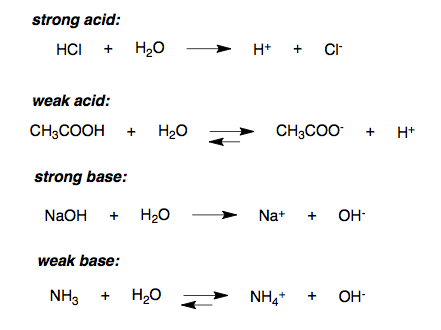



Strength Of Acids Boundless Chemistry




Easy Way To Memorize The 7 Strong Acids And 6 Strong Bases Youtube




Tips For Identifying Acid And Base Strength Concept Chemistry Video By Brightstorm



Solved Part A Classify Each Substance As A Strong Acid Chegg Com




List Of Common Strong And Weak Acids




Is Hydrofluoric Acid A Strong Or Weak Acid




Key Gcc



2



0 件のコメント:
コメントを投稿